- The most important factor that affects zeta potential is pH
- A zeta potential value quoted without a definition of it’s environment (pH, ionic strength, concentration of any additives) is a meaningless number
Why Does Zeta Potential Change?
Imagine a negative zeta potential in suspension
- If more alkali is added to this suspension then the particles tend to acquire more negative charge
- If acid is added to this suspension then a point will be reached where the charge will be neutralized
- Further addition of acid will cause a build up of positive charge
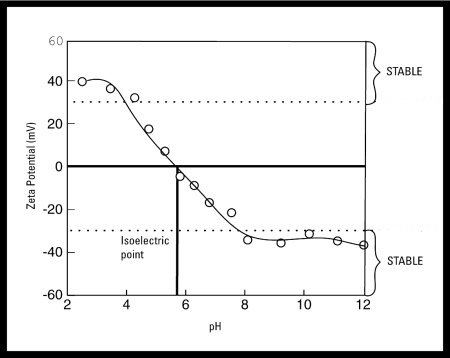
- In general, a zeta potential versus pH curve will be positive at low pH and lower or negative at high pH
- There may be a point where the curve passes through zero zeta potential
- This point is called the isoelectric point and is very important from a practical consideration
- It is normally the point where the colloidal system is least stable